


In early 2014, PRIM&R explored the topic of what we call individuals who volunteer for research in a post titled What’s in a name? Research “participant” versus research “subject.” In this post, PRIM&R concludes that, on the whole, “subject” is the most appropriate title for those involved in research studies (recognizing, however, that in some instances “participant ” may be appropriate; for example, in community-based participatory and participant-led research).
While we respect the very thorough argument presented, herein we present the contrasting points of view of three individuals: Jeff Taylor, a 35-year HIV survivor who is an HIV research advocate and has participated in many research trials and who prefers the term “volunteer;” Karine Dubé, a social scientist who has led HIV research activities and prefers the term “participant;” and Brandon Brown, an epidemiologist who has also led HIV research and argues that the term “subject” is not respectful of their contribution, though he does not have a strong preference between “volunteer” and “participant”.
As a starting point for our discussion on terminology, the Oxford dictionary defines the proposed terms as follows:
- volunteer: a person who freely offers to take part in an enterprise or undertake a task
- participant: a person who takes part in something
- subject: a person or thing that is being discussed, described, or dealt with
We also outline the two most common arguments for the use of the term “subjects” as:
- The term “participant” is not found in federal regulations governing human subjects research.
- Without agency, people who join research should not be called participants, or else we glorify unethical studies.
In response to the first argument, we point out that there many examples of laws/guidelines that were once in place but deemed unacceptable over time and subsequently modified. We recognize that changing a term in the federal guidelines could be an administrative and political nightmare, but this does not mean that it should not be changed if a need for change is determined—especially if the research community comes to a consensus.
The second argument , meanwhile, may actually expose an underling issue that leads us to use the term “subject”—”subject” assumes that people who join research do not have agency in that research. In other words, they don’t necessarily have a voice in the research being done. But perhaps studies taking place without “community voice” or the opportunity for participants to provide input are inherently unethical. And in fact, many funding agencies, including the National Institutes of Health, require involvement of communities (or at least a community advisor) when community engagement is relevant to the study.
Community voice (as there are various levels of participation) might include participants advocating for funds to undertake the project, helping plan and coordinate the project (community-based participatory research), or simply actively choosing to volunteer for the project. In recent years especially, the role of volunteers in research projects has expanded; they might serve as stakeholders, advisors, consultants, peer educators, and even act as researchers themselves. For example, in high risk/low benefit studies like HIV cure research, people living with the condition being studied can become true partners in that research—and one could argue that research should leave space for their input.
People should have a choice of participating in a study; a person is not a thing to be acted upon. We believe everyone can get on board with this statement, but how does it provide further argument against using the term subject? For an answer on that, we should ask people who participate in research, since they are the ones we are referring to in this debate. Perhaps we should practice the ethical principles of respect and autonomy, and allow them to decide what they want to be called.
In a study of 400 people living with HIV in 2015, we did just that, asking study participants the following question (348 responded, with 87% response rate):
‘If you were to participate in an HIV cure-related study, how would you describe yourself’? 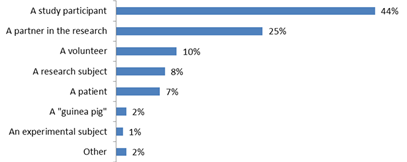
Figure 1: Response from n = 348 people living with HIV to the question: “If you were to participate in an HIV cure-related study, how would you describe yourself’?
While the question may have been biased slightly towards the term “participant”, respondents were given 8 different options of what they preferred to be called, ranging from “participant” to “guinea pig”. Nearly half of all people surveyed preferred to be called “study participants”, a quarter preferred to be called “partner”, and just 8% “research subject”. (Surprisingly, some preferred the term “guinea pig”, and we might ask ourselves what this says about their views and experience participating in research). In addition to this survey, there have been several requests from community members to use the term “participant”, including from individuals on community advisory boards who are insulted by the use of the term subject, which they say has a negative connotation and may hinder participation in research.
This is just a small snapshot of what research subjects themselves prefer—and this is only the people who were given an opportunity to speak on this topic. It is time we use more participatory methods in research, and provide people who participate in research the opportunity to be heard. Let’s ask people what they want to be called, and let’s ask them what research priorities they would like explored in their community. Without community participation, there will be no sustainability and maybe no community buy-in for research, and that’s just unethical. Lack of community input may also lead to bad study designs where people may not enroll in or be retained in research, making it a costly failure.
Critics of our argument may point out the fact that we are concentrating on HIV research, which may be very different from other types of research. We agree, but considering its well-known commitment to human rights and its history of activism, the HIV research community is ideally suited to lead the way. Plus, there are plenty of other examples where the shift from “subject” to “participant” is already happening. The NIH sponsored HIV networks (HPTN, MTN, HVTN) mandated the use of the term “participant” in their protocols and informed consent forms years ago. Other non-HIV focused sources are also moving in this direction. ClinicalTrials.gov, which reports broad clinical research, refers to people as participants, not subjects. The New England Journal of Medicine, American Journal of Public Health, and International Committee of Medical Journal Editors all use the term participant exclusively. NIH Director Francis Collins is quoted as saying, “Medical advances would not be possible without participants in clinical trials.”
With increased emphasis on patient-centered care and shared decision-making in the United States, clinical researchers need to start appreciating patients as willingly donating their time in research, instead of being “subjected” to experiments.
References
Bromley E, Mikesell L, Jones F, Khodyakov D. ‘From Subject to Participant: Ethics and the Evolving Role of Community in Health Research’. American Journal of Public Health 2015; 105(5): 900 – 908. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25790380
Brandon Brown is an assistant professor of medicine at the University of California, Riverside. Karine Dubé is an assistant professor of public health at the University of North Carolina, Chapel Hill, Gillings School of Global Public Health. Jeff Taylor is the Director of the Coachella Valley Community Research Initiative in Palm Springs, California.

I, at this point, work on health disparity research outside of HIV and can only agree– I have been told over and over from my participants that the term ‘subject’ is demeaning and “feels” bad- as if there is something done “to” them — making them passive observes/experiencers of their own lives. In my work I NEVER use the term ‘subject’– for the same reasons– and have to fight with our IRB who has a tendency to want to move us that direction. They are, however, coming around to see this our way- at least for community studies. I feel that this openness to be sensitive to not label people “subjects” should extend beyond community studies to all studies– as it takes an active decision by the person to volunteer ones body and/or thoughts around an issue or treatment.
Both sides of this debate are persuasive. In my own writing, I often go back and forth between ‘subject’ and ‘participant.’ I agree that ‘participant’ sounds more respectful than ‘subject’. And clearly, we want to treat such people–whatever we call them–with respect. However, the term ‘participant’ seems to imply a type of active involvement that is not always present. For example, if investigators are doing medical records review research, the people who are being studied have no active role. They are subjects, not participants.
As a sociologist I fully agree with using the term “participant” versus “subject.” Indeed, most sociologists refer to “research participants” and have made the switch from “research subjects” many years ago.
I think that “subjects” is impersonal, implies passivity, and emphasizes and lends itself more to a power imbalance between researchers and participants. It is important to mitigate this power imbalance as much as possible and wherever possible. One way to reduce the power imbalance between researchers and participants is recognition of the role that participants have in research.
Participants are not subservient to researchers, as the term “subject” suggests. Through their engagement and willingness to participate in research, research participants make active and tangible contributions to the research. There would be no research without willing and engaged participants. The term “participants” connotes greater acknowledgement that participants are not passively engaged. Participants give their time, often travel, exert significant mental and/or physical energy, etc.
Thus, research participants should be shown the utmost respect wherever possible and using the term “participant” is a good first step. This respect should also be followed by changes in research practice, such as sharing results with participants upon completion of the study, soliciting feedback from participants throughout their participation or as their participation concludes, as well as thanking them for their participation.
Excellent discussion that highlights the importance of language and verbage in research and science. The term that we use to refer to the individuals who participate in research studies is not just about semantics or even what appeals most to individuals to garner participation and promote recruitment. It reflects our values and philosophy as scientists. Do we see the individuals who participate in research as people (participants and volunteers) or as things (subjects)? Let’s not lose sight of why we conduct science and research in the first place- to advance knowledge in order to improve the lives of people. Thanks to the bloggers for this thoughtful post.
As one who has been in FDA trials as both a participant and investigator, I feel best about the terms participant or partner. But, throw in another term, “hero.” Similar to those who serve in the military, there is a risk of what they do, but they are participating in research that may lead to life- saving decisions. They need respect.
Please delete “subjects” these are people not animals they should be showed the respect that they are due and be referred to as participants.
Personally, I find myself using all three as inter-changeable terms. However, context does influence my preference. Almost always when communicating with lay audiences I almost always try to avoid using the term ‘subjects’. It’s painfully clinical sounding. However in certain peer-to-peer settings, I do feel like that there is an implied understanding that all three connote the same role. Folded in those terms are the same level of respect, mission, and appreciation that we are tasked to provide as members of the public health community. I’d be more concern whether researchers recognize these contextual differences and can act accordingly with the same level of sensitivity as they do with their own work.
The article, and most of the discussion, fails to distinguish between ‘subject’ and ‘object’. The research participants under discussion, and they are only some of the participants in research besides the researchers themselves, funders, reviewers, etc.) should never be treated as objects but they can be considered subjects (“subjectivity. Pertaining to the subject and his or her particular perspective, feelings, beliefs, and desires: The Oxford Companion to Philosophy, p. 857).